Abstract
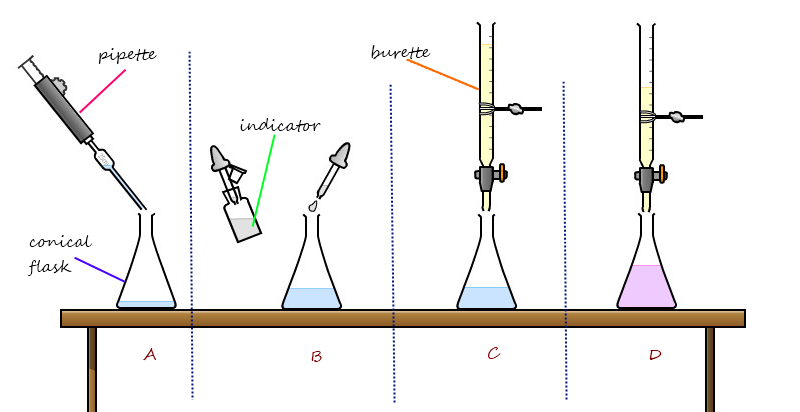
In the lab we grind the low dose and regular aspirin, after grinding the low dose aspirin we added isopropyl alcohol(5.0mL) in the grinding bowl with the low dose aspirin, then we poured the low dose aspirin and the alcohol in the flask then we put NaOH in the burette. After that we put the low dose aspirin and the isopropyl alcohol in the flask then we added 25.0mL of DI water into the flask along with the phenolphthalein indicator, then we mixed it together by slowly swirling all the ingredients that we put in the flask. Then we slowly added drops of NaOH on flask which contains the low dose of aspirin, isopropyl alcohol of 5.0mL and 25.0mL of DI water. Then as we slowly added the NaOH into the flask we waited for the chemicals to dissolved into a light pink. We found that the more we put NaOH the chemicals turns into more kind of a purple, which it was cool.
Titration is a slow addition of one solution of a known concentration to a known volume of another solution of unknown concentration until the reaction reaches neutralization, which is often indicated by the color change. Well when we crushed the aspirin which contains acid and when we added NaOH is neutralized.
Present Technology
Well pharmacists have to calculate the number of doses, and the qualities of how much a patient's needs per pill and day. Because o